 
Complete enrollment of the registration-enabling Part 2 of the PIONEER trial of AYVAKIT in non-advanced SM in mid-2021.Initiate a global Phase 1 trial of BLU-945 in patients with treatment-resistant EGFR-driven NSCLC in the second quarter of 2021. 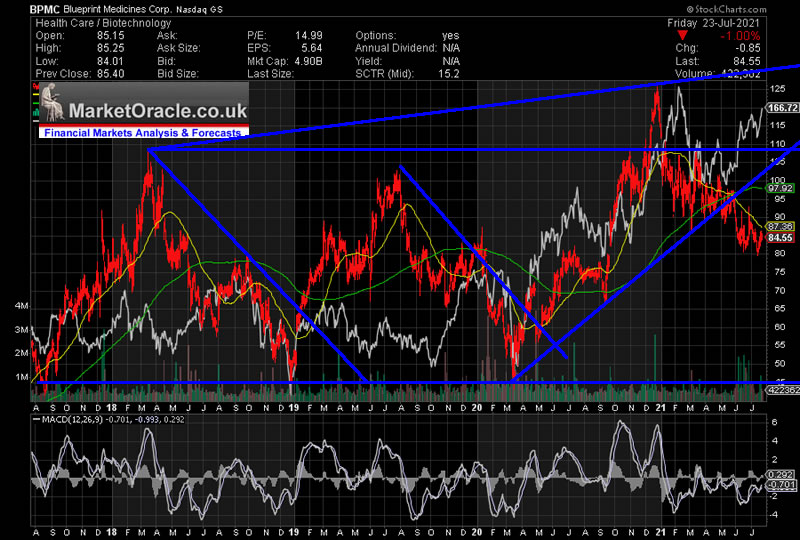
Here is a list of key upcoming trial milestones the company listed within the press release that accompanied first quarter results. Development is proceeding under multiple fronts. The company also has a diverse pipeline of which an up-to-date snapshot can be seen here. This compound is also in its commercialization infancy with just $1.8 million in net product revenues accruing to Blueprint in the first quarter of this year. This compound is marketing with developmental partner Genentech in the States and by collaboration partner CStone Pharmaceuticals in China where it has been approved for the treatment of adults with locally advanced or metastatic RET fusion-positive NSCLC after platinum-based chemotherapy. This compound was approved by the FDA in September or 2020 for the treatment of adult patients with metastatic rearranged during transfection (RET) fusion-positive non-small cell lung cancer (NSCLC) as detected by an FDA approved test and in December 2020 for the treatment of patients 12 years of age and older with advanced or metastatic RET-mutant medullary thyroid cancer and RET fusion-positive thyroid cancer. The company's second approved product is GAVRETO. Last week, the company received FDA approval around AYVAKIT for the treatment of adult patients with advanced systemic mastocytosis (Advanced SM), a rare hematologic disorder opening a potential lucrative new indication for this compound. This compound is just starting down its commercialization path and did some $7.1 million in net product sales in the first quarter. Recently, AYVAKIT/AYVAKYT was approved in China via the company's collaboration with CStone Pharmaceuticals ( OTCPK:CSPHF), for the treatment of adults with unresectable or metastatic PDGFRA exon 18 mutant GIST. It then was greenlighted by the European Commission in September of 2020 as a monotherapy for the treatment of adult patients with unresectable or metastatic GIST harboring the PDGFRA D842V mutation. This compound was approved by the FDA in January of 2020 for the treatment of adults with unresectable or metastatic GIST harboring a PDGFRA exon 18 mutation, including PDGFRA D842V mutations. The more important of these is AYVAKIT/AYVAKYT, a kinase inhibitor. 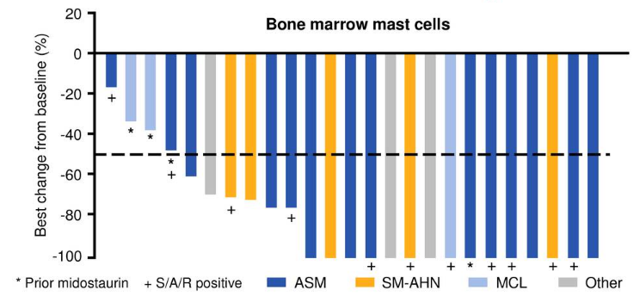
The company has two products on the market already. The stock currently trades just above $80.00 a share and sports an approximate market capitalization of just north of $5 billion.īlueprint Medicines is just starting to move into the commercialization phase. The company is focused on developing treatments in a variety of disease areas including cancer and hematologic disorders as well as multiple programs for genomically defined cancers, systemic mastocytosis, and cancer immunotherapy. Company Overview:īlueprint Medicines Corporation ( NASDAQ: BPMC) is a biotech concern based just outside of Boston in Cambridge, Massachusetts. Add in a robust balance sheet and collaboration deals with larger players, the firm merited further investigation. The company has just garnered a key FDA approval and has multiple 'shots on goal' within its developing pipeline. Today, we take an in-depth look at a mid-cap biotech concern. Life is what happens to us while we are making other plans."― Allen Saunders Marcin Klapczynski/iStock via Getty Images 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
